AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Atomic size trend ap che8/31/2023 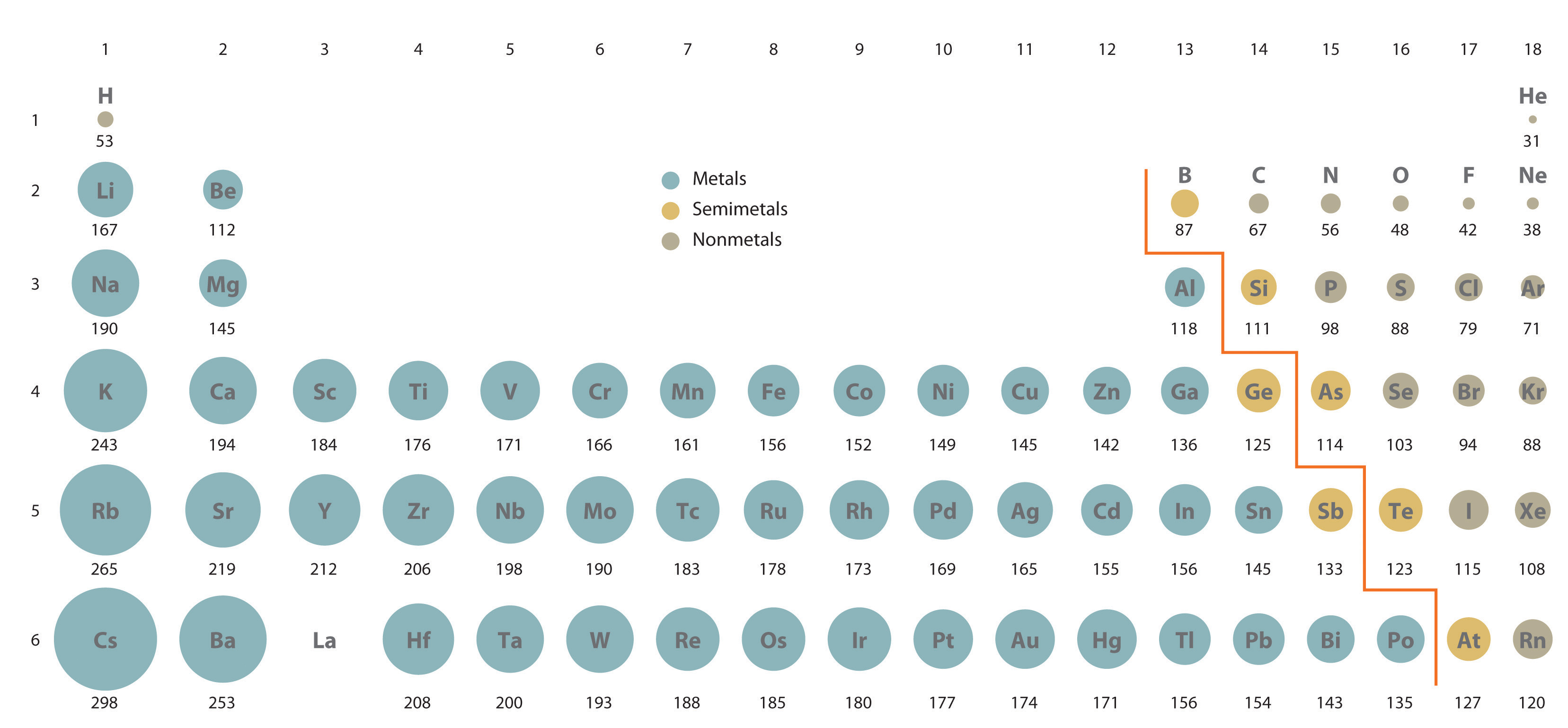 
The element that has the largest atomic radius is cesium. Which elements have the highest atomic radius? (As the number of energy levels increases, the size must increase.) Going across a period (from left to right), the number of protons increases and therefore the nuclear charge increases. The atomic size increases from the top to the bottom in any group as a result of increases in all of the three factors. As you go to the right, the atomic size trend decreases because you are adding one more proton to the nucleus (the positively-charged center of the atom) each time you move one element to the right. The chemical rationale for changes in electron affinity across the periodic table is the increased effective nuclear charge across a period and up a group.Ītomic size trend increases as you go down and to the left on the periodic table. These trends are not necessarily universal. What is the trend in EA across a period?Įlectron affinity generally increases across a period in the periodic table and sometimes decreases down a group. A periodic trend is attributed to regular variations in the atomic structure of each element. The organization of the periodic table shows the periodic trends of six different physical properties of the elements: atomic radius, electron affinity, electronegativity, ionization energy, and metallic/nonmetallic character.Ī periodic trend is a regular variation the properties of an element with increasing atomic number. 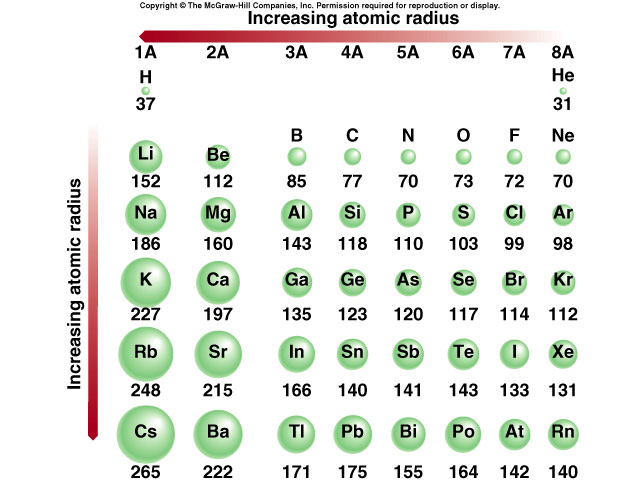
How many trends are in the periodic table? Across a period, effective nuclear charge increases as electron shielding remains constant. In general, atomic radius decreases across a period and increases down a group. What is the trend in atomic radius down a period? What Are The Trends In The Modern Periodic Table? What are the trends in modern periodic table? 
Therefore, the size of atoms decreases as one moves across a period from left to right in the periodic table. Why does atomic size decrease from left to right?Įxperiments have shown that the first case is what happens: the increase in nuclear charge overcomes the repulsion between the additional electrons in the valence level. So as the Atomic size increase so does Ionization Energy. Ionization energy increases across a period and decreases down the family. Ionization energy is the energy required to remove the most loosely held electron from a gaseous atom or ion. What are the trends for atomic size and ionization?Ītomic size is the distance from the nucleus to the valence shell. In general, the size of an atom will decrease as you move from left to the right of a certain period. How does atomic size increase on periodic table?Īn atom gets larger as the number of electronic shells increase therefore the radius of atoms increases as you go down a certain group in the periodic table of elements. This is because, within a period or family of elements, all electrons are added to the same shell.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
